Сlient
A large multinational corporation from New York used Elinext’s help to develop a compliant platform that would allow researchers to manage clinical documents.
Project
A multinational American enterprise specializing in global communications and language services attempted to improve document collaboration in clinical trials. They developed a web application and needed to update it according to the latest standards of the industry, compliances, etc.
The company hired a developer in India for that task, but the outcomes fell short of their expectations. In search of a suitable replacement, they discovered Elinext.
Challenges
The enterprise needed the system built on new technologies:
- Make sure the outdated solution based on the Domino platform will be replaced with the one with an enhanced functions set.
- Make the complete refactoring to the latest ExtJS version.
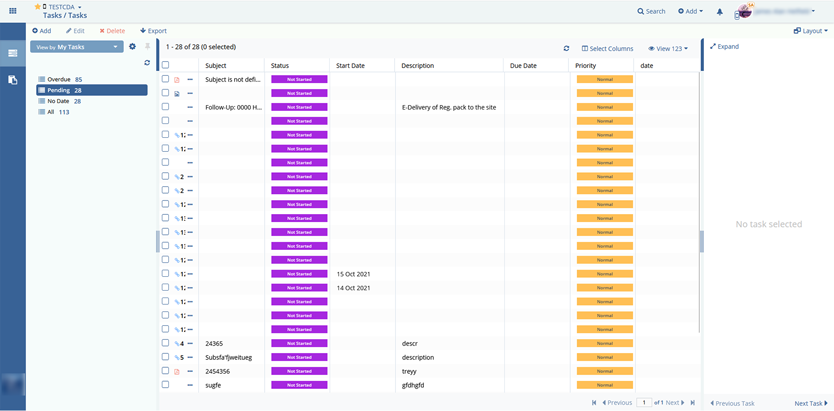
- Simplify the tasks associated with document management.
Process
Throughout the project, the Elinext team has been working in close contact with the client.
During the first two years of cooperation, our team used the waterfall methodology. As our team gradually improved the development process, we considered moving to Scrum. At that point, it was still a relatively new approach, and our client hired a scrum coach who led the transition at our premises.
As a result, the Elinext team adopted a system where each minor average release (patch) takes one month to complete. It includes in-house manual verification of the fix made and a full test automation round. For major releases that happen once a year, we run a full regression testing cycle.
We have used Skype and email for daily syncups and Microsoft Teams for planning and demo meetups. In addition, some of our team members have traveled to other countries to meet with the client and discuss further development plans once a year.
Elinext’s team size varied from 8 to 20 people during the early stages of the project. But it later grew, reaching 65 members, including backend and frontend developers, DevOps engineers, and manual and automation QAs.
At the same time, the client has employed about 200 people in-house and outsourced parts of the development all around the world.
Solution
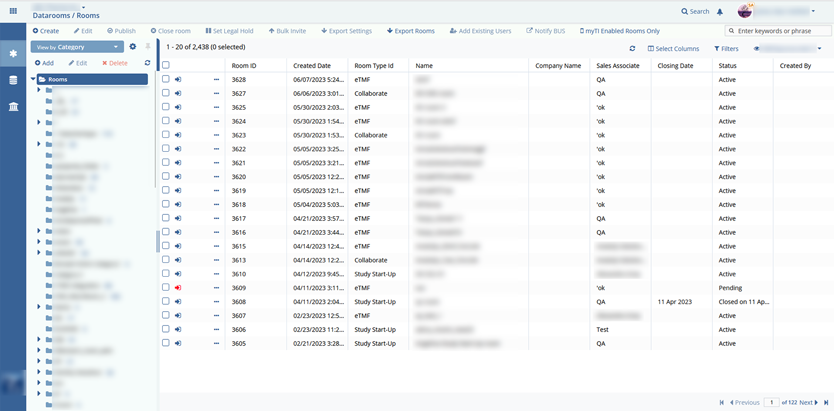
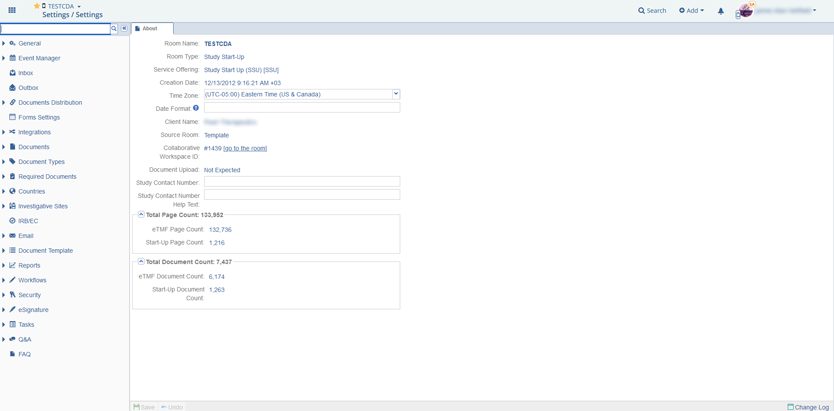
From 2010 till now, the Elinext team has been developing a fully functional SaaS platform for clinical trial management and collaboration with the configurable modular interface and securely hosting files in a CFR-compliant centralized virtual data room.
In the course of work, the team worked on the following modules:
Super Admin panel (can be outsourced to a client pharmaceutical company):
- high-level instance configuration;
- enable/disable modules.
- Study-level admin panel (for support engineers, access can be granted to client reps):
- general settings;
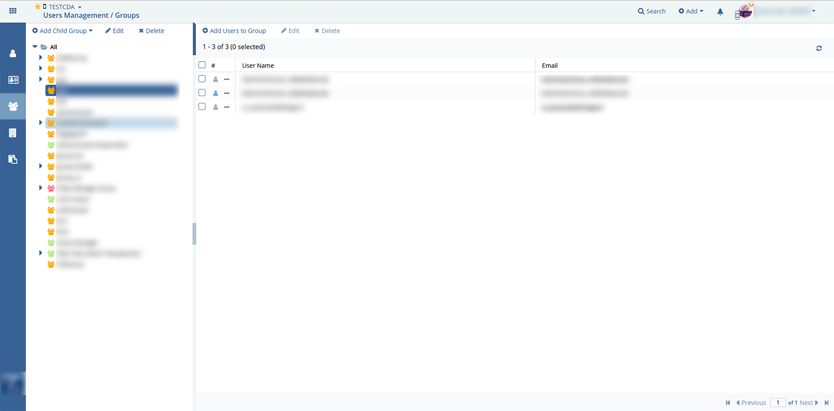
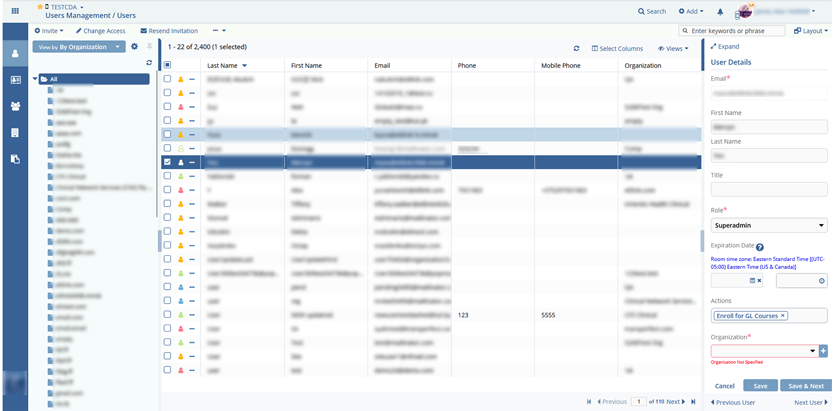
- Users & Groups management;
- configuration for document Security;
- Workflow configuration;
- document management configuration;
- eTMF studies configuration;
- Reports configuration;
- Investigative sites configuration;
- Study Start-up configuration.
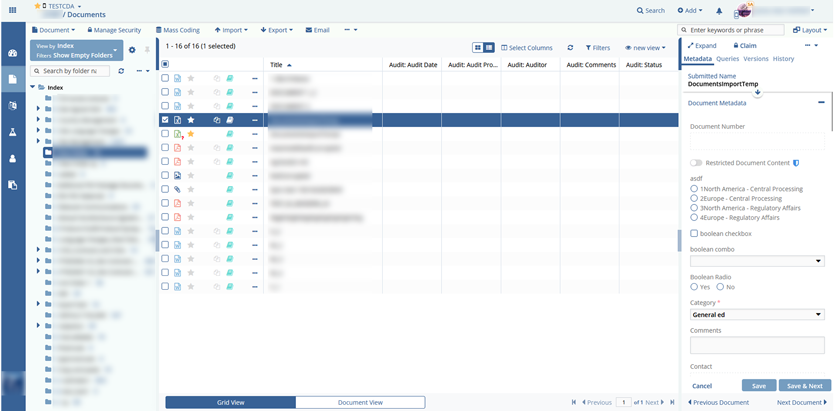
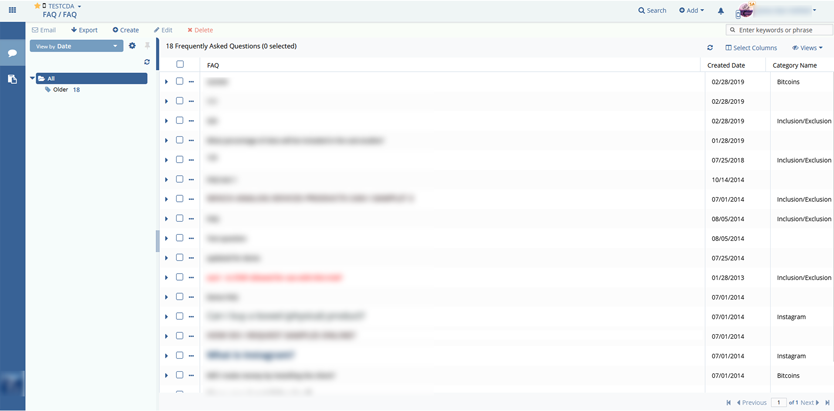
Client accounts (labs, clinics, doctors, external pharma auditors in clinical research) which included/allowed the following features:
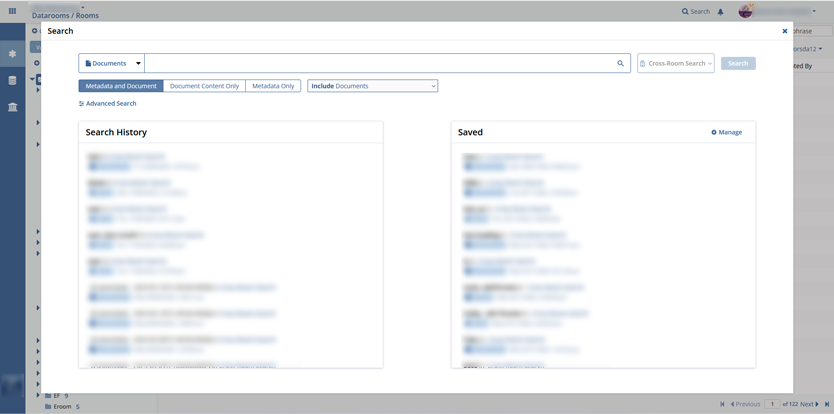
- document management;
- metadata coding;
- reports & dashboards;
- task management;
- audit;
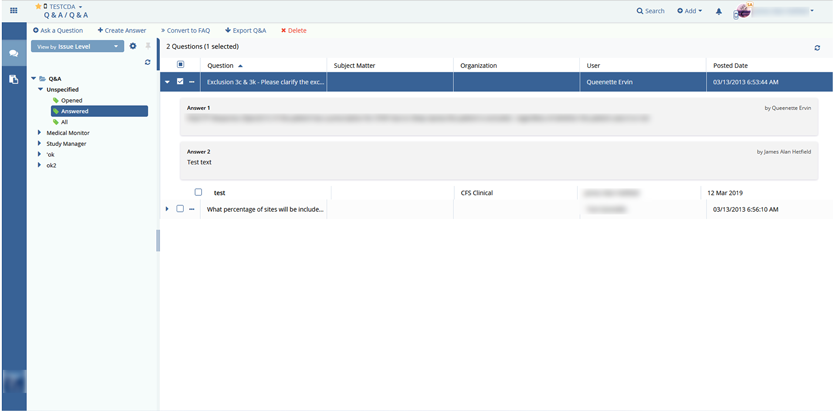
- communication;
- Q&A;
- FAQ.
The team also implemented all dashboards and metrics to review how clinical trials are held with charts, graphs, and diagrams. Using a report module, one can audit any action for the particular study.
Multiple documents can be imported to an archive or export documents’ metadata to HTML, EXCEL, or WORD files.
The system is deployed on 150 servers and has over 50,000 users. Testing documentation preparation and auditing comply with 21 CFR Part 11 guidelines.
Specific features
- Document security management with electronic signature – every user with appropriate rights can review the document with some restrictions to save, copy and etc., and sign if required.
- Automatic recognition of a document type.
- Modules migration from ExtJS-based solution to Angular.
- A lot of third-party modules integration: IRB/EC using APIs, eSignature, Alfresco, Sharepoint, DocuSign, Adlib, PleaseReview, AbobeSign, Seclore, Snowbound viewer, ARender viewer, Prizm viewer.
- Extensive integration with a new CMS system Alfresco saving all the features and benefits.
Results
We have helped our US client build a powerful application compliant with FDA regulations. It processes data in hundreds of gigabytes and has 45,000 users — professionals operating in the pharmaceutical industry and across healthcare.
Our team continues developing the project, suggesting improvements and new solutions. The next big step in this journey is creating a quality management system.
Related Case Studies
Our expertise extends beyond this project, as we have successfully delivered multiple solutions in related domains:
- QA for Clinical Trials Software – Comprehensive quality assurance services for clinical trial solutions, enhancing software reliability and regulatory compliance.
- Legal Document Review and Production App – A sophisticated application designed to optimize the document review process in the legal industry, improving accuracy and workflow efficiency.